Nature ( 2026 ) Cite this article
Escherichia coli is a leading cause of neonatal sepsis, with infection occurring in approximately one in every 1,000 live births 1 , 2 .
However, with E.
coli colonization beginning soon after birth 3 , 4 , 5 and defects in neonatal host defence maturation 6 , 7 , 8 , 9 , an alternative consideration is why infection does not occur even more frequently.
Here we show that newborn babies with E.
coli sepsis have selectively reduced vertically transferred natural antibodies that recognize E.
coli , mechanistically explaining their susceptibility to infection.
Complementary preclinical studies show that preconceptual intestinal colonization with probiotic E.
coli Nissle 1917 (EcN) 10 primes anti- E.
coli immunoglobulin G (IgG) antibodies with broad cross-reactivity to clinical isolates responsible for neonatal sepsis that override the inherent susceptibility of neonatal mice.
Outer membrane protein A (OmpA) is a target of maternal IgG and is also essential for EcN colonization-induced serological immunogenicity.
Upon vertical transfer to neonates, colonization-primed anti- E.
coli IgG uniquely protects against infection via opsonization, requiring both complement and IgG Fc receptors.
Compared with specimens from sex and gestational age-matched healthy control babies without infection, dried blood spot specimens collected one day after birth from 100 babies with E.
coli sepsis show consistently reduced IgG titres to pooled E.
coli clinical isolates and OmpA, along with impaired IgG-dependent antibacterial opsonization.
Together, these results demonstrate that natural infection susceptibility of neonates is efficiently rescued by anti- E.
coli IgG and identify defects in pathogen-targeted vertically transferred immunity as a primary risk factor for severe invasive infection in newborn babies.
Similar content being viewed by others
A live attenuated vaccine to prevent severe neonatal Escherichia coli K1 infections
Predictors of mortality and severe illness from Escherichia coli sepsis in neonates
Predictive factors for perinatal bacterial transmission from colonized mothers to delivered very-low-birth-weight infants: a retrospective cohort study
E.
coli is a pervasive Gram-negative bacterium that colonizes nearly all individuals and is an important cause of serious invasive infection 11 , 12 , 13 .
These considerations highlight unresolved questions regarding how colonization affects infection susceptibility.
For extra-intestinal infections such as urinary tract or bloodstream infections, colonization by strains that express defined virulence factors such as fimbrial adhesins or capsular polysaccharides is often identified as an infection risk factor 14 , 15 , 16 .
However, despite near ubiquitous colonization by strains capable of invasive infection, E.
coli bacteraemia still occurs infrequently, ranging from 10–100 per 100,000 person-years in the general population 17 , 18 , suggesting that host-associated factors have more important roles, especially for individuals at high risk for invasive infection.
A provocative alternative consideration relates to increasingly recognized protective benefits associated with colonization by pathobiont microorganisms.
Staphylococcus aureus bacteraemia-related death and all-cause mortality are significantly reduced among S.
aureus -colonized individuals despite higher rates of nosocomial bacteraemia 19 .
Natural antibodies—defined as those that are not related to obvious previous infection or immunization—against E.
coli and other commensal bacteria have also been consistently identified in healthy individuals 20 , 21 and mice 22 , 23 , suggesting that systemic immunogenicity primed by commensal colonization is the norm rather than the exception.
Immunogenicity to E.
coli can also be primed by colonization with other cross-reactive Gram-negative bacterial species in the larger Enterobacteriaceae family 24 , 25 .
Natural antibodies induced by cross-reactive commensals protect against systemic E.
coli and Salmonella infection 23 , and protect neonatal mice against intestinal enterotoxigenic E.
coli after vertical transfer 24 .
Reciprocally, deficiency of maternal antibodies to Group B Streptococcus is correlated with serotype-specific susceptibility of neonates to invasive infection 26 .
Although these findings suggest that defects in vertically transferred anti- E.
coli natural immunity contribute to infection susceptibility in neonates, broader applicability remains uncertain as neither immunogenicity primed by E.
coli colonization, nor antibody levels in human babies with E.
coli infection, nor susceptibility to systemic infection by extra-intestinal pathogenic E.
coli strains that cause neonatal sepsis have been evaluated.
Here we leveraged emerging platforms that enable antibody recovery from dried blood spot specimens 27 , collected one day after birth for routine newborn screening, with preclinical models of E.
coli preconceptual colonization and neonatal infection to demonstrate that early-life susceptibility reflects diminished quantity and functional opsonization activity of vertically transferred natural maternal antibodies.
EcN colonization induced immunogenicity
Mice reared under standard specific pathogen-free laboratory conditions exhibit facility-dependent variation and are often devoid of commensal E.
coli , which limits translational applicability, considering that humans are ubiquitously colonized 11 , 12 , 24 .
This shortcoming was bypassed by first establishing E.
coli colonization, followed by investigating colonization-induced immunogenicity and susceptibility to extra-intestinal infection.
We found that sustained intestinal colonization with the non-pathogenic human probiotic E.
coli Nissle 1917 (EcN) strain is efficiently established and reaches a plateau in the faeces of adult mice without antibiotic treatment, within 7 days after a single oral inoculation 10 (Fig.
1a ).
EcN colonization was limited to proximal and distal digestive organs, and spared normally sterile tissues including the spleen, liver and brain (Extended Data Fig.
1a ).
Nonetheless, colonization primed progressively increased serum anti-EcN IgG titres that plateaued by day 50 and were most enriched with IgG2b and IgG1 compared with other antibody isotypes, which were increased more modestly or remained at background levels similar to those of control mice without EcN colonization (Fig.
1b ; Extended Data Fig.
1b,c ).
Fig.
1: E.
coli Nissle (EcN) colonization induced immunogenicity and protection against invasive systemic infection.
a , EcN in the faeces after a single oral inoculation (2 × 10 6 CFU) to 6-to-8-week-old C57BL/6 wild-type mice under specific pathogen-free laboratory conditions.
b , IgG end point EcEPT titres against EcN in the sera of mice described in a .
c , Fold change in IgG titres compared with opsonization activity against the indicated E.
coli isolates in the sera of mice 50 days after EcN colonization compared with control mice without E.
coli colonization ( n = 4 individual mice (EcN, SCB61); n = 12 (UTI89); n = 3 (SCB12, SCB29, SCB34, SCB58, RS218, SCB60, SCB37)).
d , e , IgG titres that recognize five pooled OmpA outer loop peptides ( d ) compared with scramble control peptides ( e ) in mice 50 days after oral inoculation with EcN ( n = 10) or Δ ompA EcN ( n = 10) compared with control mice without E.
coli colonization ( n = 10).
f , IgG titres against EcN (left) or Δ ompA EcN (right) for mice 50 days after oral inoculation with EcN ( n = 10) or Δ ompA EcN ( n = 10) compared with control mice without E.
coli colonization ( n = 8).
g , h , Survival and bacterial burden 1 day after intravenous infection with SCB61 ( g ) or UTI89 ( h ) for mice 50 days after oral inoculation with EcN ( n = 8 ( g ); n = 7 ( h )) or control mice without E.
coli colonization ( n = 8 ( g ); n = 7 ( h )).
Data are presented as mean ± s.d.
Differences between groups were analysed using simple linear regression ( c ), one-way ANOVA ( d – f ), unpaired Student’s t- test ( g , h ) or log-rank (Mantel–Cox) test ( g , h ).
LOD, limit of detection.
Source data Full size image
a , EcN in the faeces after a single oral inoculation (2 × 10 6 CFU) to 6-to-8-week-old C57BL/6 wild-type mice under specific pathogen-free laboratory conditions.
b , IgG end point EcEPT titres against EcN in the sera of mice described in a .
c , Fold change in IgG titres compared with opsonization activity against the indicated E.
coli isolates in the sera of mice 50 days after EcN colonization compared with control mice without E.
coli colonization ( n = 4 individual mice (EcN, SCB61); n = 12 (UTI89); n = 3 (SCB12, SCB29, SCB34, SCB58, RS218, SCB60, SCB37)).
d , e , IgG titres that recognize five pooled OmpA outer loop peptides ( d ) compared with scramble control peptides ( e ) in mice 50 days after oral inoculation with EcN ( n = 10) or Δ ompA EcN ( n = 10) compared with control mice without E.
coli colonization ( n = 10).
f , IgG titres against EcN (left) or Δ ompA EcN (right) for mice 50 days after oral inoculation with EcN ( n = 10) or Δ ompA EcN ( n = 10) compared with control mice without E.
coli colonization ( n = 8).
g , h , Survival and bacterial burden 1 day after intravenous infection with SCB61 ( g ) or UTI89 ( h ) for mice 50 days after oral inoculation with EcN ( n = 8 ( g ); n = 7 ( h )) or control mice without E.
coli colonization ( n = 8 ( g ); n = 7 ( h )).
Data are presented as mean ± s.d.
Differences between groups were analysed using simple linear regression ( c ), one-way ANOVA ( d – f ), unpaired Student’s t- test ( g , h ) or log-rank (Mantel–Cox) test ( g , h ).
LOD, limit of detection.
To investigate cross-reactivity to other E.
coli strains, and particularly to clinically relevant isolates, sera from EcN-colonized mice were titred against a panel of E.
coli clinical isolates.
Given the importance of sequence types 95, 69, 131 and 1193 for causing neonatal sepsis 28 , 29 , 30 , we focused on clinical isolates of these sequence types, which have distinct O-antigen, K-capsule and flagella serotypes that were non-overlapping with EcN 29 , 31 , 32 , 33 , 34 , 35 , 36 (Extended Data Fig.
1d ).
EcN colonization primed broad cross-reactivity with significantly increased IgG titres against each isolate in the sera of EcN-colonized mice compared with in control mice without EcN colonization (Extended Data Fig.
1e ).
Sera from EcN-colonized mice also efficiently opsonized these clinical isolates, with opsonization activity directly proportional to the IgG titre (Fig.
1c ), highlighting functional activity against virulent E.
coli by natural antibodies primed by commensal EcN.
To further investigate E.
coli cross-reactive antigens, these analyses were extended to a panel of isogenic strains with targeted mutations in genes encoding serotype-defining cell-surface structures, including waaL (encoding a ligase required for O-antigen attachment to lipid A core), kpsD (encoding a transporter required for K-capsule presentation), fliC (encoding a major flagella structural protein) or rfaH (encoding a transcription elongation factor required for O-antigen and K-capsule expression) 37 , 38 .
Despite loss of these serotype-defining antigens individually or in combination, cross-reactivity was preserved with similar increased anti- E.
coli IgG in the sera of EcN-colonized mice when titred against the mutant strains compared with their respective parental wild-type strain (Extended Data Fig.
2 ).
Other targets of host immunity include ubiquitous outer membrane proteins, such as OmpA, which possesses multiple transmembrane domains and four outer surface loop domains 35 , 39 (Extended Data Fig.
3a ).
OmpA immunogenicity was probed using five peptides, encompassing the outer loop 1, 2 and 4 domains plus two distinct loop 3 peptides, given the wider heterogeneity in these residues between EcN and clinical isolates that cause invasive infection (Fig.
1d and Extended Data Fig.
3a,b ).
EcN colonization induced sharply increased anti-OmpA loop peptide-specific IgG, which was overturned in mice colonized with Δ ompA EcN (Fig.
1d and Extended Data Fig.
3b,c ), and without cross-reactivity to scramble control peptides (Fig.
1e ).
Of note, sera from Δ ompA EcN-colonized mice exhibit reduced wild-type EcN-specific IgG and lack Δ ompA EcN-specific IgG, indicating that OmpA is not only a target, but is also essential for EcN-primed serological immunogenicity (Fig.
1f ).
However, serological immunogenicity primed by commensal EcN is not exclusively targeted against OmpA, as titres against Δ ompA EcN were still consistently increased (Fig.
1f and Extended Data Fig.
4 ).
In turn, nearly identical increased IgG titres were found against other Δ ompA variants of established clinical isolates compared with their respective parental wild-type strain (Extended Data Fig.
4 ).
These findings align with serological cross-reactivity primed by EcN colonization to other Gram-negative Enterobacteriaceae with distinct OmpA outer loop peptides including Klebsiella , Yersinia , Citrobacter , Enterobacter and Salmonella species, but not against unrelated microorganisms, including Gram-positive S.
aureus and Listeria monocytogenes bacteria or Candida albicans fungi (Extended Data Fig.
5a,b ).
Together, these findings identify OmpA as one of several serological targets for natural antibodies primed by commensal E.
coli and as a necessity for E.
coli colonization-induced immunogenicity.
Protective anti- E.
coli natural antibody
Protection primed by EcN colonization was evaluated by comparing susceptibility of mice infected intravenously with virulent E.
coli .
We focused particularly on SCB61, which represents a strain with pronounced EcN cross-reactivity, and UTI89, which has reduced cross-reactivity and is widely used in preclinical studies 6 , 31 (Fig.
1c and Extended Data Fig.
1e ).
These experiments showed significantly improved survival after infection with either SCB61 or UTI89, and reciprocally reduced bacterial burden in target tissues of EcN-colonized mice compared with in those of control mice without EcN colonization (Fig.
1g,h and Extended Data Fig.
6a ).
Improved survival and reduced pathogen burden were more pronounced after SCB61 infection than after UTI89 infection, consistent with relative levels of EcN serological cross-reactivity (Fig.
1g,h and Extended Data Figs.
1e and 6a ).
Comparatively, protection against UTI89 infection was abolished in Δ ompA EcN-colonized mice lacking cross-reactive IgG (Extended Data Fig.
6b,c ), in agreement with the necessity for OmpA in priming immunogenicity.
Significantly reduced pathogen burden in EcN-colonized mice after infection with Klebsiella pneumoniae , but not L.
monocytogenes , S.
aureus or C.
albicans , (Extended Data Fig.
6d–g ) further highlights protection by cross-reactive antibodies, rather than trained innate immune components 40 .
To more definitively establish the necessity for antibodies in colonization-induced protection, susceptibility to invasive infection was evaluated in mice transferred with serum from EcN-colonized wild-type or B cell-deficient μMT −/− mice, or from wild-type control donors without EcN colonization.
These experiments showed that antibodies are essential for colonization-induced protection, as only sera from EcN-colonized wild-type mice were able to transfer protection (Fig.
2a and Extended Data Fig.
6h ).
The importance of antibodies is further shown by the lack of EcN colonization-induced protection against UTI89 infection in μMT −/− mice compared with in wild-type mice (Fig.
2b and Extended Data Fig.
6i ).
Lack of protection in μMT −/− mice cannot be explained by inefficient EcN colonization given the similar colonization tempo and EcN recovery in the faeces (Extended Data Fig.
6j ).
Together, these findings demonstrate that commensal E.
coli colonization primes natural antibodies that protect against invasive systemic infection.
Fig.
2: Protective natural anti- E.
coli maternal antibodies override neonatal infection susceptibility.
a , Bacterial burden 1 day after SCB61 (left) or UTI89 (right) infection for mice administered with sera (100 μl) from control mice without EcN colonization ( n = 8), wild-type (WT) mice on day 50 after EcN colonization ( n = 8) or μMT −/− mice ( n = 7) on day 50 after EcN colonization.
b , Bacterial burden 1 day after UTI89 infection in wild-type mice compared with μMT −/− mice on day 50 after EcN colonization ( n = 12 (wild type); n = 13 ( μMT −/− )) or control mice without E.
coli colonization ( n = 12 (wild type); n = 11 ( μMT −/− )).
c , Bacterial burden 1 day after UTI89 infection (200 CFU intraperitoneal inoculation) in 3-day-old pups born to control mice without E.
coli colonization ( n = 9) compared with pups born to EcN-colonized wild-type ( n = 10) or EcN-colonized μMT −/− ( n = 5) dams.
d , Bacterial burden 1 day after UTI89 infection in 3-day-old pups administered the indicated dosage of purified IgG from EcN-colonized or control mice without E.
coli colonization 1 h prior to infection ( n = 3 to 5 mice per group; each dot represents data from an individual mouse).
e , Bacterial burden 1 day after UTI89 infection (left) or compared with anti-EcN IgG titres (right) for pups born to EcN-colonized dams ( n = 10), pups born to control mice without E.
coli colonization ( n = 9) or cross-fostered within the first 12 h after birth ( n = 9 (prenatal); n = 8 (postnatal)).
f – h , Human anti- E.
coli IgG levels in paired third trimester maternal specimens compared with cord blood after term pregnancy, titred against EcN ( f ), pooled neonatal infection clinical isolates RS218, SCB12, SCB29, SCB61, SCB34, SCB58, SCB37 and SCB60 ( g ) or a pool containing five OmpA outer loop peptides compared with scramble control peptides ( h ) ( n = 10; lines indicate paired maternal–cord blood specimens).
Data are presented as mean ± s.d.
Differences between groups were analysed using one-way ANOVA ( a , c , e (left)), unpaired Student’s t -test ( b ), simple linear regression ( e (right)) or paired Student’s t -test ( f – h ).
Source data Full size image
a , Bacterial burden 1 day after SCB61 (left) or UTI89 (right) infection for mice administered with sera (100 μl) from control mice without EcN colonization ( n = 8), wild-type (WT) mice on day 50 after EcN colonization ( n = 8) or μMT −/− mice ( n = 7) on day 50 after EcN colonization.
b , Bacterial burden 1 day after UTI89 infection in wild-type mice compared with μMT −/− mice on day 50 after EcN colonization ( n = 12 (wild type); n = 13 ( μMT −/− )) or control mice without E.
coli colonization ( n = 12 (wild type); n = 11 ( μMT −/− )).
c , Bacterial burden 1 day after UTI89 infection (200 CFU intraperitoneal inoculation) in 3-day-old pups born to control mice without E.
coli colonization ( n = 9) compared with pups born to EcN-colonized wild-type ( n = 10) or EcN-colonized μMT −/− ( n = 5) dams.
d , Bacterial burden 1 day after UTI89 infection in 3-day-old pups administered the indicated dosage of purified IgG from EcN-colonized or control mice without E.
coli colonization 1 h prior to infection ( n = 3 to 5 mice per group; each dot represents data from an individual mouse).
e , Bacterial burden 1 day after UTI89 infection (left) or compared with anti-EcN IgG titres (right) for pups born to EcN-colonized dams ( n = 10), pups born to control mice without E.
coli colonization ( n = 9) or cross-fostered within the first 12 h after birth ( n = 9 (prenatal); n = 8 (postnatal)).
f – h , Human anti- E.
coli IgG levels in paired third trimester maternal specimens compared with cord blood after term pregnancy, titred against EcN ( f ), pooled neonatal infection clinical isolates RS218, SCB12, SCB29, SCB61, SCB34, SCB58, SCB37 and SCB60 ( g ) or a pool containing five OmpA outer loop peptides compared with scramble control peptides ( h ) ( n = 10; lines indicate paired maternal–cord blood specimens).
Data are presented as mean ± s.d.
Differences between groups were analysed using one-way ANOVA ( a , c , e (left)), unpaired Student’s t -test ( b ), simple linear regression ( e (right)) or paired Student’s t -test ( f – h ).
IgG overrides neonatal susceptibility
Given pronounced early-life susceptibility and rising incidence of E.
coli as a cause of severe infection in newborn babies 1 , 2 , this platform was used to further investigate whether preconceptual maternal EcN colonization can override neonatal susceptibility to E.
coli infection.
Neonatal mice are markedly more susceptible to relatively low inocula of UTI89 (200 colony-forming units (CFU)) administered by intraperitoneal inoculation than adult mice (Extended Data Fig.
7a ).
Despite this inherent early-life susceptibility, mortality and high tissue pathogen burden were reduced in 3-day-old pups born to EcN-colonized dams compared with age-matched control pups born to dams without EcN colonization (Fig.
2c and Extended Data Fig.
7b ).
Protection in neonatal mice requires maternal antibodies, as mortality and pathogen burden rebound in μMT +/− pups born to EcN-colonized μMT −/− dams, and to levels resembling those in pups born to dams without EcN colonization (Fig.
2c and Extended Data Fig.
7b ).
Protection was similarly observed after oral E.
coli infection in pups born to EcN-colonized dams, representing a more relevant route of infection leading to systemic dissemination 41 (Extended Data Fig.
7c ).
Anti- E.
coli IgG and IgG subclasses were efficiently transferred with similar titres in EcN-colonized dams and their offspring, compared with selectively reduced IgM in pups (Extended Data Fig.
7d ), in agreement with inefficient placental transfer of this isotype 42 .
Colonization-induced IgG antibodies are also sufficient for protection, given the reduced pathogen burden in neonatal mice administered low-dosage IgG from EcN-colonized donors compared with mice without E.
coli colonization (Fig.
2d and Extended Data Fig.
7e ).
Vertically transferred protection is also not explained by commensal E.
coli in-host evolution 43 , given the near-identical genomes of EcN recovered from colonized virgin or pregnant mice compared with the parental strain (Extended Data Fig.
7f ).
Thus, preconceptual E.
coli colonization primes natural maternal antibodies that, upon vertical transfer, efficiently protect neonates against invasive systemic infection.
To further investigate when protective anti- E.
coli IgG transfer occurs, pups born to EcN-colonized dams or to non-colonized control dams were cross-fostered within the first 12 h after birth, with infection occurring 3 days thereafter.
These experiments showed progressively reduced susceptibility in pups with only prenatal exposure compared with those with only postnatal exposure, which were both significantly less susceptible than control pups with neither prenatal nor postnatal exposure to EcN-colonized dams (Fig.
2e and Extended Data Fig.
7g ).
Although these findings show that protection is transferred both in utero and after birth, postnatal exposure through breastmilk has a more prominent role, given the only modestly increased bacterial burden and marginally reduced anti-EcN IgG titres compared with pups born to and nursed by EcN-colonized dams (Fig.
2e and Extended Data Fig.
7g ).
Applicability to human mother–infant dyads was evaluated by comparing anti- E.
coli titres in paired third trimester maternal and cord blood specimens after term pregnancy (Extended Data Fig.
8a ).
This analysis showed similar anti-EcN IgG titres, including individual IgG subclasses, in cord blood compared with maternal blood specimen, whereas anti-EcN IgM titres declined precipitously (Fig.
2f and Extended Data Fig.
8b ).
IgG with specificity to E.
coli clinical isolates that cause neonatal sepsis and OmpA were also efficiently transferred, as shown by similar titres against a mixture of the aforementioned eight neonatal sepsis clinical isolates containing two of each sequence type (Fig.
2g and Extended Data Fig.
1d ), or OmpA loop peptides individually or pooled together in paired maternal–cord blood specimens compared with significantly reduced background titres against scramble control peptides (Fig.
2h and Extended Data Fig.
8c ).
Notably, as cord blood enables the contribution of in utero transfer to be evaluated separately and dissociated from transfer via breastmilk, these findings also demonstrate more efficient prenatal transfer of natural maternal IgG in human babies than in mouse pups, which are more reliant on postnatal acquisition of protective antibodies through breastmilk 44 , 45 .
IgG protects neonates via opsonization
To investigate how vertically transferred natural antibodies confer protection, we evaluated the effects of adoptively transferred serum from EcN-colonized adult donor mice on infection susceptibility in neonatal recipients.
These experiments showed sharply reduced bacterial burden in E.
coli -infected pups transferred with sera from EcN-colonized donors compared with sera from control mice without EcN colonization, and an essential role for antibodies, as protection was eliminated in pups transferred with sera from EcN-colonized μMT −/− donors (Fig.
3a and Extended Data Fig.
8d ).
This versatile platform was expanded to evaluate the requirement for defined antibody-interacting molecules by comparing how pups with targeted defects in these molecules respond to sera from EcN-colonized donors.
These experiments show that complement and IgG Fc receptors (FcγR), including FcγRI, FcγRII, FcγRIII and FcγRIV 46 , are simultaneously essential, as protection conferred by sera from EcN-colonized wild-type donors is eliminated in FcγR null , C3 −/− and C1q −/− pups (Fig.
3b and Extended Data Fig.
8e ).
By contrast, CD22, which binds to sialylated glycoproteins including IgG 47 , is non-essential given the similar protection in Cd22 −/− pups and in wild-type pups (Fig.
3b and Extended Data Fig.
8e ).
Fig.
3: Natural anti- E.
coli antibodies selectively protect neonates through opsonization.
a , Bacterial burden 1 day after UTI89 infection (200 CFU, intraperitoneal inoculation) in 3-day-old pups administered sera from control mice without EcN colonization ( n = 8) compared with sera from EcN-colonized wild-type ( n = 5) or EcN-colonized μMT −/− ( n = 8) donors.
b , Bacterial burden 1 day after UTI89 infection in 3-day-old pup or eight-week-old adult FcγR null , C3 −/− , C1q −/− or Cd22 −/− recipients administered sera from EcN-colonized wild-type donor mice ( n = 6 to 8 mice per group; each dot represents data from one mouse) compared with sera from control mice without E.
coli colonization ( n = 6 to 8 mice per group; each dot represents data from one mouse).
c , EcN opsonization by sera from control mice without EcN colonization ( n = 7) compared with sera from wild-type ( n = 7), μMT −/− ( n = 7) or C3 −/− ( n = 7) EcN-colonized donors in RAW264.7 mouse macrophage cells.
d , EcN opsonization by sera from control mice without E.
coli colonization ( n = 6) compared with sera from EcN-colonized donors ( n = 6) with anti-CD16 and anti-CD32 Fc blockade (anti-CD16/32) or rat IgG2b isotype control antibody in RAW264.7 mouse macrophage cells.
e , Opsonization of E.
coli (pooled mix of neonatal clinical isolates RS218, SCB12, SCB29, SCB61, SCB34, SCB58, SCB37 and SCB60) by cord blood sera from term pregnancy after heat inactivation, supplementation with IgG-depleted human sera, or with anti-human Fc block, compared with goat polyclonal IgG isotype control antibody in THP1 activated human macrophage cells (left) or HL60 human differentiated neutrophil cells (right) ( n = 8 unique cord blood specimens per group (THP1 cells); n = 6 unique cord blood specimens per group (HL60 cells)).
Data are presented as mean ± s.d.
Differences between groups were analysed using one-way ANOVA ( a , c – e ) or unpaired Student’s t -test ( b ).
Source data Full size image
a , Bacterial burden 1 day after UTI89 infection (200 CFU, intraperitoneal inoculation) in 3-day-old pups administered sera from control mice without EcN colonization ( n = 8) compared with sera from EcN-colonized wild-type ( n = 5) or EcN-colonized μMT −/− ( n = 8) donors.
b , Bacterial burden 1 day after UTI89 infection in 3-day-old pup or eight-week-old adult FcγR null , C3 −/− , C1q −/− or Cd22 −/− recipients administered sera from EcN-colonized wild-type donor mice ( n = 6 to 8 mice per group; each dot represents data from one mouse) compared with sera from control mice without E.
coli colonization ( n = 6 to 8 mice per group; each dot represents data from one mouse).
c , EcN opsonization by sera from control mice without EcN colonization ( n = 7) compared with sera from wild-type ( n = 7), μMT −/− ( n = 7) or C3 −/− ( n = 7) EcN-colonized donors in RAW264.7 mouse macrophage cells.
d , EcN opsonization by sera from control mice without E.
coli colonization ( n = 6) compared with sera from EcN-colonized donors ( n = 6) with anti-CD16 and anti-CD32 Fc blockade (anti-CD16/32) or rat IgG2b isotype control antibody in RAW264.7 mouse macrophage cells.
e , Opsonization of E.
coli (pooled mix of neonatal clinical isolates RS218, SCB12, SCB29, SCB61, SCB34, SCB58, SCB37 and SCB60) by cord blood sera from term pregnancy after heat inactivation, supplementation with IgG-depleted human sera, or with anti-human Fc block, compared with goat polyclonal IgG isotype control antibody in THP1 activated human macrophage cells (left) or HL60 human differentiated neutrophil cells (right) ( n = 8 unique cord blood specimens per group (THP1 cells); n = 6 unique cord blood specimens per group (HL60 cells)).
Data are presented as mean ± s.d.
Differences between groups were analysed using one-way ANOVA ( a , c – e ) or unpaired Student’s t -test ( b ).
A similar approach comparing susceptibility of adult recipient mice transferred with sera from EcN-colonized donors was used to investigate whether this necessity for complement and FcγR is unique to the neonatal developmental window.
Notably, and in sharp contrast to 3-day-old recipients, protection by sera from EcN-colonized mice persisted in adult 8-week-old FcγR null , C3 −/− and C1q −/− recipient mice (Fig.
3b and Extended Data Fig.
8e ).
These findings unveil developmental distinctions in how natural immunity works, with IgG being selectively reliant on opsonization through FcγR and complement in neonates.
Together with diminished early-life complement levels and neutrophil activity 48 , 49 , these results further highlight that neonates have minimal excess capacity in these immune components compared with adults, with more substantial functional redundancy among protective antibody-interacting molecules.
Anti- E .
coli IgG in human neonatal sepsis
To further investigate whether E.
coli infection in human neonates reflects inadequate vertically transferred natural maternal antibodies, anti- E.
coli IgG titre and opsonization activity were evaluated in babies with neonatal sepsis utilizing banked dried blood spot specimens collected the first day after birth (median 25 h) for routine newborn screening (Fig.
4a ).
Babies with E.
coli sepsis were identified retrospectively based on International Classification of Disease (ICD) diagnostic codes and were compared with three control babies without infection who were individually matched for sex, gestational age (±1 week) and birth timing (same month and year) to normalize these variables potentially affecting vertically transferred immunity (Fig.
4a ).
Specimens from 100 babies with neonatal E.
coli sepsis showed similar sex distribution, with a wide distribution in gestational age (median 35 weeks, range 23 to 42 weeks) and infection age (median 4 days, range 1 to 29 days) (Fig.
4b ).
Although not a criterion used for selection or exclusion, none of these babies had galactosaemia, a known risk factor for E.
coli sepsis that is identified by newborn screening 51 .
Given the limited sample quantity and lack of information on particular E.
coli features that cause infection in this retrospective platform, together with the established importance of E.
coli sequence types 95, 69, 131 and 1193 in human neonatal sepsis 28 , 29 , 30 , anti- E.
coli activity was evaluated using a mixture of the aforementioned 8 neonatal sepsis clinical isolates containing 2 of each sequence type (Extended Data Fig.
1d ).
Fig.
4: Diminished anti- E.
coli IgG and opsonization activity in newborn blood spot specimens from babies with neonatal sepsis.
a , Dried newborn blood spot specimens were obtained from babies with E.
coli neonatal sepsis and three control babies without infection who were matched for sex, gestational age and infection timing.
Left, photo courtesy of Chris Lima, March of Dimes.
Middle, BioTrust logo reprinted with permission from MDHHS Communications.
Right, ELISA plate schematic created in BioRender; Way, S.
S.
https://biorender.com/wz9mo0w (2026).
b , Birth gestational age and infection onset age distribution among 100 babies with neonatal E.
coli sepsis.
c , Anti- E.
coli IgG titres in newborn blood spots from babies with E.
coli sepsis ( n = 100) compared with the average in matched control babies ( n = 100) titred with pooled neonatal infection clinical isolates (RS218, SCB12, SCB29, SCB61, SCB34, SCB58, SCB37 and SCB60) evaluated by birth gestational age.
d , IgG levels in newborn blood spots from babies described in c ( n = 100 ( E.
coli sepsis); n = 100 (averaged control babies)).
e , f , IgG titres in newborn blood spots against five pooled OmpA loop peptides ( e ) or scramble control peptides ( f ) for babies described in c ( n = 100 ( E.
coli sepsis); n = 100 (averaged control babies)).
g , h , Anti- E.
coli opsonization activity for newborn blood spot specimens against eight pooled neonatal infection clinical isolates in THP1 activated macrophage cells ( g ) or HL60 differentiated neutrophil cells ( h ) ( n = 100 ( E.
coli sepsis); n = 100 (averaged control babies)).
Each dot represents data from an individual blood spot specimen from babies with E.
coli sepsis (red) or the average from three matched control babies for each case (blue); the shaded region indicates 95% confidence intervals.
Data are mean ± s.d.
Differences between groups analysed using unpaired Student’s t -test ( b , gestational weeks), Mann–Whitney U test ( b , infection onset age), paired Student’s t -test ( c – h ), or regression intercepts using analysis of covariance ( c – h ).
Source data Full size image
a , Dried newborn blood spot specimens were obtained from babies with E.
coli neonatal sepsis and three control babies without infection who were matched for sex, gestational age and infection timing.
Left, photo courtesy of Chris Lima, March of Dimes.
Middle, BioTrust logo reprinted with permission from MDHHS Communications.
Right, ELISA plate schematic created in BioRender; Way, S.
S.
https://biorender.com/wz9mo0w (2026).
b , Birth gestational age and infection onset age distribution among 100 babies with neonatal E.
coli sepsis.
c , Anti- E.
coli IgG titres in newborn blood spots from babies with E.
coli sepsis ( n = 100) compared with the average in matched control babies ( n = 100) titred with pooled neonatal infection clinical isolates (RS218, SCB12, SCB29, SCB61, SCB34, SCB58, SCB37 and SCB60) evaluated by birth gestational age.
d , IgG levels in newborn blood spots from babies described in c ( n = 100 ( E.
coli sepsis); n = 100 (averaged control babies)).
e , f , IgG titres in newborn blood spots against five pooled OmpA loop peptides ( e ) or scramble control peptides ( f ) for babies described in c ( n = 100 ( E.
coli sepsis); n = 100 (averaged control babies)).
g , h , Anti- E.
coli opsonization activity for newborn blood spot specimens against eight pooled neonatal infection clinical isolates in THP1 activated macrophage cells ( g ) or HL60 differentiated neutrophil cells ( h ) ( n = 100 ( E.
coli sepsis); n = 100 (averaged control babies)).
Each dot represents data from an individual blood spot specimen from babies with E.
coli sepsis (red) or the average from three matched control babies for each case (blue); the shaded region indicates 95% confidence intervals.
Data are mean ± s.d.
Differences between groups analysed using unpaired Student’s t -test ( b , gestational weeks), Mann–Whitney U test ( b , infection onset age), paired Student’s t -test ( c – h ), or regression intercepts using analysis of covariance ( c – h ).
These experiments showed consistent tenfold reductions of anti- E.
coli IgG in blood spots from babies with neonatal sepsis compared with the average titre in control babies without infection who were matched for sex, gestational age and birth timing (Fig.
4c ).
Reductions in anti- E.
coli IgG primarily reflect diminished IgG2 titres, which showed even more exaggerated (more than 40-fold) reductions between specimens from babies with E.
coli sepsis compared with specimens from control babies without infection, whereas anti- E.
coli IgG1 did not differ significantly, and IgG3 and IgG4 titres were only inconsistently detected (Extended Data Fig.
9a ).
These reductions in anti- E.
coli IgG are not explained by diminished overall antibody recovery, given the similar gestational age-dependent changes in total IgG and IgG subclasses between babies with E.
coli sepsis and control babies without infection (Fig.
4d and Extended Data Fig.
9b,c ).
Blood spots from babies with neonatal sepsis also contain reduced anti-OmpA IgG, albeit of lower magnitude than with detection using whole E.
coli bacteria (Fig.
4c,e ), in agreement with the aforementioned preclinical data highlighting OmpA as an important target for E.
coli serological immunogenicity.
Use of scrambled control peptides confirmed the specificity of neonatal IgG for binding to OmpA outer loop peptides (Fig.
4f ).
Given the importance of natural antibody opsonization for host defence in neonates, opsonization activity was further evaluated in these newborn blood spot specimens.
As our focus was investigating opsonization differences due to anti- E.
coli antibodies, IgG-depleted human sera containing complement was supplemented so that potential changes in opsonization activity could be directly attributed to differences in antibodies that recognize E.
coli (Fig.
3e ).
These experiments showed consistently diminished anti- E.
coli opsonization activity in macrophage and neutrophil cells for specimens from babies with E.
coli sepsis compared with the average activity in specimens from matched control babies without infection across gestational ages (Fig.
4g,h ).
Stratifying anti- E.
coli titres and IgG-dependent opsonization activity across birth gestational age further clarifies that overlap in absolute titres and opsonization activity between babies with E.
coli sepsis and control babies without infection primarily reflect reductions in overall IgG levels in babies born preterm, shown by the significant reductions in anti- E.
coli and anti-OmpA IgG titres and opsonization activity across gestational ages (Fig.
4c–h ).
Risk reduction cut-offs for each assay were also identified using conditional logistic regression to evaluate the distribution of titres and opsonization activity without consideration for gestational age (Extended Data Fig.
10a–d ).
Particularly for this cohort, which contains 100 babies with E.
coli neonatal sepsis and 296 matched control babies without infection, anti-EcN IgG end point titres (EcEPTs) below 2,500 or anti-OmpA EcEPTs below 1,000 would predict around 20% risk of neonatal sepsis (Extended Data Fig.
10a–d ), which is significantly higher than current epidemiological estimates 1 , 2 of around 1 case per 1,000 live births or 0.1% risk.
Postnatal age of infection onset within the neonatal window is another parameter that is consistently related to E.
coli susceptibility 1 , 2 .
Evaluating this parameter as an independent variable showed that reductions in anti- E.
coli IgG, anti-OmpA IgG or IgG-dependent opsonization activity, along with similar overall IgG levels and titres against anti-OmpA scramble control peptides, are maintained in babies with E.
coli sepsis regardless of age at infection onset (Extended Data Fig.
10e–j ).
For babies with early-onset sepsis, diminished anti- E.
coli IgG titre and opsonization were also not explained by antibody adsorption to bacteria as shown by comparable magnitude reductions in anti- E.
coli , anti-OmpA IgG titre and opsonization activity in babies with E.
coli sepsis identified within the first two days after birth compared with infection identified after three days of age (Extended Data Fig.
10k ).
Together with the efficiency whereby vertically transferred maternal IgG overrides the susceptibility of neonatal mice to E.
coli infection, these findings demonstrate diminished vertically transferred natural immunity in the form of anti- E.
coli antibodies and their opsonization function as universal risk factors for neonatal sepsis.
Neonates are highly susceptible to E.
coli , with severe infections occurring in up to 1 in every 1,000 live births 1 , 2 .
However, infections might be expected to occur even more frequently given ubiquitous exposure with transfer of pathogenic maternal strains shortly after birth 3 , 4 , 5 , 52 , 53 , together with hypo-responsiveness and skewed tissue distribution of neonatal immune cells 6 , 7 , 8 , 9 .
Our studies investigating why most babies do not develop E.
coli sepsis further amplify the translational importance of previous studies investigating E.
coli immunity using preclinical infection models by verifying the necessity of vertically transferred antibodies that recognize E.
coli 23 , 24 .
We find newborn blood spots from human babies with E.
coli sepsis have significantly reduced anti- E.
coli IgG titres and IgG-dependent anti- E.
coli opsonization activity (Fig.
4c–h ) that parallel the susceptibility of neonatal mice born to dams devoid of commensal E.
coli colonization-induced IgG (Fig.
2c and Extended Data Fig.
7b ).
Restoring anti- E.
coli IgG through vertical transfer from E.
coli- colonized dams or horizontal transfer from E.
coli -colonized donors efficiently protects neonatal mice against infection (Figs.
2c and 3a ), resembling the tenfold increase in anti- E.
coli IgG titre found in sex and gestation age-matched babies without infection (Fig.
4c ).
Vertical IgG transfer increases throughout gestation, peaking in the third trimester, and early preterm birth is a significant risk factor for neonatal E.
coli infection 1 , 2 , 54 .
This progressive transfer of maternal IgG, previously detected primarily in cord blood, was recapitulated in our analysis of blood spots from babies on the basis of gestational age (Fig.
4d and Extended Data Fig.
9c ).
Although total IgG levels were generally lower among infants born before 32 weeks of gestation, those with E.
coli sepsis still had reduced anti- E.
coli IgG and opsonization activity compared to gestational age-matched control individuals regardless of gestational age at birth (Fig.
4c–h ).
Isolated deficiency in anti- E.
coli IgG, rather than diminished total IgG levels, as the primary risk factor for neonatal sepsis further suggests that systemic invasion occurs much more frequently, with clinical infection developing primarily in babies without sufficient vertically transferred anti- E.
coli IgG to eliminate invasive bacteria.
In turn, levels of anti- E.
coli IgG or IgG-dependent opsonization below defined thresholds shortly after birth can be used to identify babies at highest risk regardless of gestational age at birth (Extended Data Fig.
10a–d ).
Indeed, the lower anti- E.
coli IgG levels in babies with E.
coli sepsis suggests the possibility that defects may exist in preconceptual maternal colonization, colonization-primed serological immunity, vertical transfer of these natural protective maternal antibodies or a combination of these factors.
Although individually these defects are likely to occur infrequently, their combined frequency is expected to be approximately 1 in 1,000, similar to the proportion of newborns who develop E.
coli sepsis.
These results support a general adoption of prenatal screening for anti- E.
coli IgG similar to current evaluations of expecting mothers for serological immunity against pathogens such as tetanus or varicella, to which babies are particularly vulnerable.
Targeting E.
coli OmpA, and especially outer domains of this transmembrane protein that is found in all E.
coli , may be especially informative given the reduced titres to these defined antigens in blood spot specimens from babies with E.
coli sepsis (Fig.
4e ).
Comparing anti-OmpA titres in nearly 400 blood spot specimens (100 from babies with E.
coli sepsis and 296 from matched control babies without infection) identifies levels of anti-OmpA and anti- E.
coli IgG along with IgG-dependent anti- E.
coli opsonization above which risk of sepsis is significantly decreased (Extended Data Fig.
10a–d ).
Important next steps include comparing anti-OmpA and anti- E.
coli IgG in mothers of babies with E.
coli sepsis, along with levels of these antibodies in paired maternal–neonate samples, similar to studies on Group B Streptococcus 26 .
Our finding that antibodies that recognize E.
coli are prevalent in healthy babies regardless of gestational age, together with preclinical data showing that anti- E.
coli IgG efficiently protects neonatal mice, whether delivered parenterally or through breastmilk, also raises the exciting prospect of preventing infection in infants at high risk using immunoglobulin enriched with anti- E.
coli antibodies administered systemically or via early-life feeding.
Averting infection with immunoglobulin may extend to other pathogens responsible for neonatal sepsis, including K.
pneumoniae , given the reduced IgG with this specificity associated with neonatal sepsis 55 .
Finally, considering broad cross-reactivity primed by EcN colonization against E.
coli clinical isolates that cause neonatal sepsis, exploring use of this probiotic strain in women of reproductive age or expecting mothers to reduce E.
coli neonatal infections, similar to the way in which maternal immunization against influenza, COVID-19 or tetanus can reduce neonatal infection 56 , 57 , 58 , 59 , represents compelling actionable next steps.
Unlike subunit vaccines that are currently being developed for E.
coli , which prime immunity against a limited set of defined antigens 35 , 60 , immunogenicity primed by commensal EcN expressing a more diverse array of endogenous protective antigens with cross-reactivity to virulent strains regardless of O-antigen, K-capsule and flagella serotypes would be expected to confer broader protection.
Alternatively, given the OmpA immunogenicity associated with protection against systemic infection in both preclinical infection models and natural infection of babies resulting in neonatal sepsis, further investigations on the use of OmpA and other E.
coli immunodominant outer membrane proteins as candidate vaccine antigens are warranted.
Inherent limitations associated with retrospective identification of neonatal E.
coli sepsis and associated use of de-identified dried blood spot specimens collected for newborn screening include the lack of maternal prenatal specimens and microbiological data on the exact E.
coli strain that caused sepsis in each individual, and whether infected neonates had other immunological or anatomic risk factors.
These considerations prevent evaluation of maternal E.
coli colonization as well as strain-specific virulence and antigenic features of pathobiont E.
coli , which should also be a focus of follow-up studies to further investigate the link with diminished vertically transferred IgG in neonatal E.
coli sepsis.
Additional next steps for realizing these translational considerations include prospectively evaluating these parameters in larger human maternal–infant birth cohorts that are sufficient to capture the approximate 1 case in around 1,000 live births.
Nonetheless, reframing how neonatal E.
coli infection is conceptualized, focusing on resistance instead of susceptibility for the majority of babies, and incorporating critical elements related to vertically transferred natural maternal immunity yields insights into unique mediators of early-life immunity with important implications for understanding protection against other human pathobionts.
Wild-type mice on the C57BL/6 background and transgenic strains including µMT −/− (B6.129S2- Ighm tm1Cgn /J; 002288), Cd22 −/− (C57BL/6- Cd22 tm1Lam /J; 006940), C3 −/− (B6.129S4- C3 tm1Crr /J; 029661) and C1q −/− (B6(Cg)- C1qa tm1d(EUCOMM)Wtsi /TennJ; 031675) were purchased from The Jackson Laboratory 61 , 62 , 63 , 64 and maintained under specific pathogen-free housing conditions at Cincinnati Children’s Hospital.
FcγR null (FcRα null) mice with combined defects in FcγRI, FcγRII, FcγRIII, FcγRIV were provided by J.
Ravetch 46 .
All mice were housed under specific pathogen-free conditions in individually ventilated cages with ad libitum food (tekland 2919) and ultrafiltered UV-sterilized water under controlled environmental conditions including 22 °C, humidity 30–80% and 14 h–10 h light–dark daily cycles.
Sex and age-matched groups of adult (6–8 weeks) mice were randomized for EcN colonization or no colonization control groups, and monitored for up to 150 days thereafter.
For cross-fostering, pregnant mice were checked for delivery at least every 12 h, and pups were transferred to dams with or without EcN colonization after birth.
Infected mice were checked at least every 12 h and euthanized when moribund for survival outcome experiments.
All animal studies were performed under Cincinnati Children’s Hospital Research Foundation IACUC approved protocols.
Microbial colonization and infection
Features for E.
coli strains Nissle 1917 (EcN), UTI89, RS218, SCB12, SCB29, SCB61, SCB34, SCB58, SCB37 and SCB60 are described in Extended Data Fig.
1d .
EcN and other parental wild-type E.
coli strains including EC958, MS7163 and RS218 (refs.
65 , 66 , 67 ; Extended Data Fig.
4a ) and mutants with targeted defects in serotype-defining cell-surface structures ( waaL , kpsD , fliC , rfaH or ompA ) were generated by λ-Red-mediated homologous recombination as described 37 , 38 , 68 .
Reference wild-type K.
pneumoniae (43816), Yersinia enterocolitica (JB580v), Citrobacter koseri (BAA-895), Enterobacter cloacae (13047), Salmonella typhimurium (ST14028), L.
monocytogenes (10403 s), S.
aureus (USA200) and C.
albicans (SC5314) have been described 69 , 70 , 71 , 72 , 73 , 74 , 75 .
For oral inoculation, EcN was cultured overnight in brain–heart infusion (BHI) liquid medium, sub-cultured to early log-phase growth (OD 600 0.1; 37 °C; 200 rpm), washed and diluted in sterile saline and administered dropwise into the mouth of unanaesthetized adult mice (2 × 10 6 CFU in 50 µl).
For infection, bacterial strains were cultured overnight in BHI liquid medium, sub-cultured to early log-phase growth (OD 600 0.1; 37 °C; 200 rpm), washed and diluted in sterile saline and administered intravenously via the lateral tail vein.
Adult mice were infected with E.
coli (5 × 10 6 CFU for bacterial burden quantification; 4 × 10 7 CFU for survival outcomes).
Three-day-old neonatal mice were infected with E.
coli (200 CFU in 50 µl, intraperitoneally or 5 × 10 6 CFU, oral gavage) as described 6 , 76 .
Other bacteria including K.
pneumoniae , L.
monocytogenes and S.
aureus were administered at the following dosages intravenously (1 × 10 5 CFU K.
pneumoniae ; 1 × 10 5 CFU L.
monocytogenes ; 7.5 × 10 6 CFU S.
aureus in 200 µl) for infection in adult mice.
For fungal infection, C.
albicans was cultured overnight in yeast extract peptone adenine hemisulfate dextrose (YPAD) liquid medium, sub-cultured early log-phase growth (OD 600 0.1; 30 °C; 200 rpm), washed and diluted in sterile saline and administered intravenously to adult mice (1 × 10 6 CFU) as described 77 .
The bacterial and fungal inoculum in each experiment was verified by spreading serial dilutions onto media agar plates.
For enumerating the number of recoverable E.
coli , K.
pneumoniae , L.
monocytogenes , S.
aureus or C.
albicans from colonized or infected mice, the faeces or tissue was dissected in sterile conditions and homogenized in saline containing 0.05% Triton X-100.
Serial dilutions of the faeces or tissue homogenates were spread onto media agar plates, and the number of individual colonies enumerated after incubation at 37 °C for 24 h.
Blood collected from adult EcN-colonized or control mice via retroorbital or cheek bleeding, or from neonatal mice after decapitation, was allowed to clot at room temperature for 20 min and then centrifuged for serum collection.
For adoptive transfer, sera from donor mice were pooled and transferred intraperitoneally to 8-week-old adult (100 µl) or 3-day-old neonatal (50 µl) recipient mice 4 h prior to infection.
For IgG purification, frozen serum was thawed and purified using NAb Protein A Plus Spin Column (Thermo Fisher Scientific), quantified on the basis of optical density (NanoDrop), verified for purity by protein gel electrophoresis and Coomassie Blue staining, resuspended in sterile saline and administered intraperitoneally to 3-day-old neonatal recipient mice 1 h prior to infection.
Paired maternal and cord blood specimens after term pregnancy were collected through the IMPRINT cohort designed to investigate the effect of initial respiratory virus exposures on infant immunity 78 .
This cohort began in October 2019 after receiving Institutional Review Board approvals from Cincinnati Children’s Hospital Medical Center and the University of Cincinnati Medical Center (IRB 2019-0629 and SITE00000489, respectively).
Women were enroled in the third trimester of pregnancy, with collection of maternal sera and cord blood collected at delivery.
Additional non-paired cord blood specimens from healthy donors were provided in a de-identified manner through Cincinnati Children’s Hospital Applied Gene and Cell Therapy Center, Cell Processing Facility.
For newborn blood spots, 100 babies with neonatal sepsis were identified through International Classification of Diseases diagnostic codes ICD9-03842 and ICD10-P36.4 corresponding to ‘ E.
coli septicemia’ and ‘sepsis of the newborn due to E.
coli ’, respectively.
ICD9-03842-based identification was limited to individuals ≤28 days old to identify neonatal infection (Fig.
4b ).
Three control babies without infection were individually matched for each case with identical sex, gestational age (±1 week) and birth timing (same month and year), except for two babies with E.
coli sepsis born at 23 weeks of gestation, where only one matched control baby could be identified, were identified through the Michigan BioTrust for Health 79 .
Three 6-mm circular punches from dried blood specimens for each baby with E.
coli sepsis and corresponding control babies without infection were provided in a de-identified manner.
All studies were performed under Cincinnati Children's Hospital Medical Center and Michigan Department of Health and Human Services IRB approved protocols.
Enzyme-linked immunosorbent assay
Antibodies against E.
coli and other bacteria and fungi were titred using high-binding 96-well immunoassay plates (Corning, 9018) coated with microorganisms from log-phase (OD 600 0.5–0.7) growth, washed and diluted in saline and allowed to dry overnight under UV light.
Thereafter, bacteria or fungi-coated plates were blocked with milk (3%), serum titred starting at a 1:10 dilution with 5 additional 1:5 serial dilutions and probed with biotin-conjugated secondary antibodies including: rat anti-mouse IgG (ThermoFisher 13-4013-85 (RRID AB_466650); 1:2,000 dilution), rat anti-mouse IgM (ThermoFisher 13-5890-82 (RRID AB_466761); 1:2,000 dilution), rat anti-mouse IgA (ThermoFisher 13-5994-82 (RRID AB_466863); 1:2,000 dilution), rat anti-mouse IgE (BD Pharmingen 553419 (RRID AB_394850); 1:2,000 dilution), rat anti-mouse IgG1 (BD Pharmingen 553441 (RRID AB_394861); 1:2,000 dilution), rat anti-mouse IgG2b (BD Pharmingen 553393 (RRID AB_394831); 1:2,000 dilution), rabbit anti-mouse IgG2c (ThermoFisher SA5-10235 (RRID AB_2810193); 1:2,000 dilution) and rat anti-mouse IgG3 (BD Pharmingen 553401; RRID AB_394838; 1:2,000 dilution).
For human anti- E.
coli antibodies, maternal or cord blood serum specimens were titred starting at a 1:10 dilution with 5 additional 1:5 serial dilutions and probed with biotin-conjugated secondary antibodies including: goat anti-human IgM (ThermoFisher A18841 (RRID AB_2535618); 1:2,000 dilution), mouse anti-human IgG1 (BD Pharmingen 555869 (RRID AB_396187); 1:2,000 dilution), rabbit anti-human IgG2 (ThermoFisher MA5-27931 (RRID AB_2744977); 1:2,000 dilution), mouse anti-human IgG3 (ThermoFisher MH1532 (RRID AB_2539711); 1:2,000 dilution) and mouse anti-human IgG4 (BD Pharmingen 555882 (RRID AB_396194); 1:2,000 dilution).
Overall human IgG levels were evaluated by pooling antibodies against each IgG subclass at 1:2,000 dilution.
For dried blood spots, each 6-mm punch, estimated to contain 5 μl of serum, was dissolved overnight at room temperature in 100 µl of sterile saline with gentle shaking 80 , 81 , 82 and titred starting at a 1:10 dilution with 5 additional 1:5 serial dilutions.
Total human antibody levels were measured using the following commercial kits according to the manufacturer’s recommendations: human IgG (Thermo Fisher Scientific, 88-50550), human IgM (Thermo Fisher Scientific, 88-50620-88), human IgG1 (Thermo Fisher Scientific, 88-50560-22), human IgG2 (Thermo Fisher Scientific, 88-50570-22), human IgG3 (Thermo Fisher Scientific, 88-50580-22) and human IgG4 (Thermo Fisher Scientific, 88-50590-22).
Plates were developed with streptavidin–peroxidase (BD Bioscience 554066; 1:1,000 dilution) using o -phenylenediamine dihydrochloride as a substrate and measuring absorbance at 450 nm.
Titres were quantified as end point titre (EcEPT), the reciprocal of the highest analyte dilution that gives a reading above the cut-off, which was set to 0.1 using a non-linear second order polynomial regression as described 47 .
For evaluating antibodies against OmpA, each peptide (loop 1, WSQYHDTGFINNNGPTHEN; loop 2, GRMPYKGSVENGAYKA; loop 3a, KSNVYGKN; loop 3b, KANVPGGASFKD; loop 4, TNNIGDAHTIGTRPDNGM; loop 1 scramble, TQPNSHNDINTNHGYGWE; loop 2 scramble, RSMAGKGPKNAGYVYE; loop 3a scramble, KYNGVNSK; loop 3b scramble, FKAKDNGVGSPA; loop 4 scramble, MTNTHNTAIGDRNIGPGD) at >90% purity (GenScript) was diluted in saline for coating 96-well (600 ng per well; for titrating mouse sera) or 384-well (120 ng per well; for titrating human sera or eluted dry blood spots) ELISA plates overnight at 4 °C.
For probing the response to multiple peptides, equal amounts of each peptide, 600 ng per well (96-well pates) or 120 ng per well (384-well plates), were utilized.
Thereafter, peptide-coated plates were blocked with 2% PBSA, and serum or eluted dry blood spots were titred starting at a 1:10 dilution with 5 additional 1:5 serial dilutions and probed with the aforementioned biotin-conjugated anti-mouse IgG and anti-human IgG secondary antibodies.
To enumerate cell-associated bacteria, E.
coli strains were transformed with GFP-expressing plasmid pBbB5c-GFP (Addgene, 35353) 83 and maintained in medium supplemented with chloramphenicol (20 µg ml −1 ).
For opsonization by mouse sera, RAW 264.7 mouse macrophage (ATCC TIB-71) cells (2 × 10 5 cells per well) were seeded in flat-bottom 96-well cell culture plates (Cellstar, 655180) and allowed to adhere for 1 h achieving >80% confluency.
Thereafter, the medium was exchanged with 30 µl of saline containing mouse sera at a 1:5 dilution plus GFP + E.
coli (10 6 CFU per well) and allowed to incubate at 37 °C for 30 min.
After incubation, the plate was washed three times with sterile saline to eliminate non-adherent bacteria and macrophage-adherent bacteria were measured by fluorescence (OD 530 after 480 nm excitation).
For some experiments, the GFP + E.
coli serum mixture was supplemented with rat anti-mouse CD16/32 (clone 2.4G2; BioXcell BE0307 (RRID AB_2736987)) or rat IgG2b kappa isotype control (clone LTF.2; BioXcell BE0090 (RRID AB_1107780)).
For opsonization by human sera or blood spot specimens, THP1 activated macrophage (ATCC TIB-202) cells (1 × 10 4 cells per well), cells were seeded into 384-well tissue culture treated plates (Corning, 3701) to >80% confluency in DMEM medium supplemented with recombinant human TGFβ1 (1 ng ml −1 ; R&D, 7754-BH) and 1α,25-dihydroxyvitamin D 3 (50 nM; Millipore Sigma, 705942) for 24 h.
Adherent cells were then stimulated with phorbol-myristate acetate (15 ng ml −1 ; Sigma-Aldrich) for 15 min for macrophage differentiation as described 84 .
For neutrophil cells, human myeloid leukaemia HL60 (ATCC CCL-240) cells (1 × 10 4 cells per well) were seeded into 384-well tissue culture treated plates (Corning, 3701) in RPMI 1640 medium supplemented with l -glutamine, penicillin–streptomycin, HEPES, 10% FBS and 1.3% DMSO for 5 days as described 85 .
Thereafter the medium was exchanged with 8 µl of saline containing human serum or blood spot elution at a final 1:40 dilution plus pooled GFP + E.
coli (2 × 10 5 total CFU per well), which were allowed to incubate at 37 °C for 30 min.
After incubation, the plate was washed three times with sterile saline to eliminate non-adherent bacteria, and adherent bacteria were measured by fluorescence (OD 530 after 480 nm excitation).
RAW 264.7, THP1 and HL60 were routinely tested (about every six months) for mycoplasma negativity.
Besides confirmation based on cellular morphology and species-specific phagocytic function after incubation with mouse or human antibodies, no additional cell line authentication was performed.
To inactivate complement, serum was heat-inactivated (56 °C, 30 min) prior to use.
For some experiments, the serum:bacteria mix was supplemented with 20% immunoglobin depleted human serum as a source of complement (Celprogen, HS2001).
Bacterial whole-genome sequencing and comparative analysis
Bacterial isolates were sequenced using a hybrid approach combining Illumina short-read sequencing (paired-end) and Oxford Nanopore long-read sequencing (Plasmidsaurus).
Raw reads were quality-assessed using FastQC (v.0.12.1) and NanoPlot (v.1.46.0) 86 .
Hybrid de novo assembly was performed using Unicycler (v.0.5.1), which utilizes SPAdes for initial short-read assembly followed by long-read scaffolding and polishing with Racon (v.1.5.0) 87 , 88 , 89 .
Assembly quality was assessed using QUAST (v.5.2.0.2) against the E.
coli Nissle 1917 reference genome (GCF_000714595.1) 90 .
Core genome alignment and single-nucleotide polymorphism (SNP) calling were performed using Parsnp (v.1.5.6) 91 .
Pairwise SNP distances were calculated from the resulting VCF file using custom Python scripts.
SNPs were annotated with gene information by mapping positions to the reference genome GFF3 annotation, and short read-based (Illumina) analysis was also performed using breseq to call mutations against the parental EcN stock genome sequence 92 .
OmpA comparative sequence analysis
OmpA protein sequences were retrieved from NCBI using the Biopython Entrez module and combined with locally derived OmpA sequences from clinical E.
coli isolates (SCB12, SCB29, SCB34, SCB37, SCB58, SCB60 and SCB61) and reference strains (EcN, RS218 and UTI89).
Multiple sequence alignment was performed using MAFFT (v.7.526) with the autoalignment function enabled 93 .
Five peptide regions corresponding to OmpA outer loops were defined based on the RS218 reference sequence: loop 1, WSQYHDTGFINNNGPTHEN; loop 2, GRMPYKGSVENGAYKA; loop 3a, KSNVYGKN; loop 3b, KANVPGGASFKD; loop 4, TNNIGDAHTIGTRPDNGM as described 35 .
Peptides were mapped to aligned sequences using MAFTT.
Peptide positions were mapped from the ungapped reference to the gapped alignment coordinates, and corresponding regions were extracted directly from all aligned sequences for comparison.
All statistical analyses were performed with Prism (v.10.4.1, GraphPad) and Python (v.12.3).
For comparing antibody titres or opsonization activity across birth gestation age or infection onset timing, significance of differences between the distribution of data from specimens from babies with E.
coli sepsis and from control babies without infection were evaluated using one-way analysis of covariance (ANCOVA).
For probability of sepsis predictions, distribution of IgG titres and opsonization activity for each baby with E.
coli sepsis and for control babies without infection individually matched for sex, gestational age and infection timing were evaluated using conditional logistic regression.
Predicted probabilities of sepsis across antibody or opsonization activity levels relative to baseline rate for cases were calculated from the fitted model, with 95% confidence intervals estimated by bootstrap resampling of matched sets (1,000 iterations).
The sample size per group is shown by individual data points that represent results from individual mice.
Where applicable, recipient mice were randomized for administration of sera from EcN-colonized wild-type or µMT −/− donors.
The distribution of data in each group was first evaluated using the D’Agostino–Pearson normality test.
For data where all groups contain normally distributed data, significance of differences between groups was evaluated using two-sided one-way ANOVA test (three or more groups to adjust for multiple comparisons) or two-sided Student’s t -test (two groups).
For data where one or more groups contains data that is not normally distributed, significance of differences between groups was evaluated using two-sided non-parametric Kruskal–Wallis one-way ANOVA test (three or more groups to adjust for multiple comparisons) or Mann–Whitney U test (two groups).
Where applicable, paired analysis between two groups was used for comparison and indicated by a connecting line between data points.
Correlation between antibody levels compared with opsonization activity, bacterial burden or gestation age were compared using simple linear regression.
Differences in survival between two groups were evaluated using the log-rank (Mantel–Cox) test.
For each analysis, P < 0.05 was defined as statistically significant and P values for key groups are indicated in the figures.
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Primary data for main and extended data figures are provided as accompanying source data and are available at https://github.com/Way-Lab/ecoli_natural_immunity .
Source data are provided with this paper.
Custom Python scripts used for SNP distance calculation, annotation and visualization are available at https://github.com/Way-Lab/ecoli_natural_immunity .
Stoll, B.
J.
et al.
Early-onset neonatal sepsis 2015 to 2017, the rise of Escherichia coli , and the need for novel prevention strategies.
JAMA Pediatr.
174 , e200593 (2020).
Article PubMed PubMed Central Google Scholar
Biondi, E.
et al.
Epidemiology of bacteremia in febrile infants in the United States.
Pediatrics 132 , 990–996 (2013).
Secher, T., Brehin, C.
& Oswald, E.
Early settlers: which E.
coli strains do you not want at birth?
Am.
J.
Physiol.
311 , G123–G129 (2016).
Al-Balawi, M.
& Morsy, F.
M.
Prenatal versus postnatal initial colonization of healthy neonates’ colon ecosystem by the enterobacterium Escherichia coli .
Microbiol.
Spectr.
9 , e0037921 (2021).
Backhed, F.
et al.
Dynamics and stabilization of the human gut microbiome during the first year of life.
Cell Host Microbe 17 , 852 (2015).
Article CAS PubMed Google Scholar
Elahi, S.
et al.
Immunosuppressive CD71 + erythroid cells compromise neonatal host defence against infection.
Nature 504 , 158–162 (2013).
Article ADS CAS PubMed PubMed Central Google Scholar
Kollmann, T.
R., Kampmann, B., Mazmanian, S.
K., Marchant, A.
& Levy, O.
Protecting the newborn and young infant from infectious diseases: lessons from immune ontogeny.
Immunity 46 , 350–363 (2017).
Olin, A.
et al.
Stereotypic immune system development in newborn children.
Cell 174 , 1277–1292.e14 (2018).
Article CAS PubMed PubMed Central Google Scholar
Araujo David, B.
et al.
Kupffer cell reverse migration into the liver sinusoids mitigates neonatal sepsis and meningitis.
Sci.
Immunol.
9 , eadq9704 (2024).
Sonnenborn, U.
Escherichia coli strain Nissle 1917-from bench to bedside and back: history of a special Escherichia coli strain with probiotic properties.
FEMS Microbiol.
Lett.
363 , fnw212 (2016).
Kaper, J.
B., Nataro, J.
P.
& Mobley, H.
L.
Pathogenic Escherichia coli .
Nat.
Rev.
Microbiol.
2 , 123–140 (2004).
Tenaillon, O., Skurnik, D., Picard, B.
& Denamur, E.
The population genetics of commensal Escherichia coli .
Nat.
Rev.
Microbiol.
8 , 207–217 (2010).
Sawhney, S.
S.
et al.
Gut microbiome evolution from infancy to 8 years of age.
Nat.
Med.
31 , 2004–2015 (2025).
Hunstad, D.
A.
& Justice, S.
S.
Intracellular lifestyles and immune evasion strategies of uropathogenic Escherichia coli .
Annu.
Rev.
Microbiol.
64 , 203–221 (2010).
Biggel, M.
et al.
Horizontally acquired papGII-containing pathogenicity islands underlie the emergence of invasive uropathogenic Escherichia coli lineages.
Nat.
Commun.
11 , 5968 (2020).
Mu, A.
et al.
Integrative omics identifies conserved and pathogen-specific responses of sepsis-causing bacteria.
Nat.
Commun.
14 , 1530 (2023).
Bonten, M.
et al.
Epidemiology of Escherichia coli bacteremia: a systematic literature review.
Clin.
Infect.
Dis.
72 , 1211–1219 (2021).
Feldman, S.
F.
et al.
A nationwide population-based study of Escherichia coli bloodstream infections: incidence, antimicrobial resistance and mortality.
Clin.
Microbiol.
Infect.
28 , 879.e1–879.e7 (2022).
Wertheim, H.
F.
et al.
Risk and outcome of nosocomial Staphylococcus aureus bacteraemia in nasal carriers versus non-carriers.
Lancet 364 , 703–705 (2004).
Cohen, I.
R.
& Norins, L.
C.
Natural human antibodies to gram-negative bacteria: immunoglobulins G, A, and M.
Science 152 , 1257–1259 (1966).
Article ADS CAS PubMed Google Scholar
Haas, A.
et al.
Systemic antibody responses to gut commensal bacteria during chronic HIV-1 infection.
Gut 60 , 1506–1519 (2011).
Koch, M.
A.
et al.
Maternal IgG and IgA antibodies dampen mucosal T helper cell responses in early life.
Cell 165 , 827–841 (2016).
Zeng, M.
Y.
et al.
Gut microbiota-induced immunoglobulin G controls systemic infection by symbiotic bacteria and pathogens.
Immunity 44 , 647–658 (2016).
Zheng, W.
et al.
Microbiota-targeted maternal antibodies protect neonates from enteric infection.
Nature 577 , 543–548 (2020).
Hofstra, H.
& Dankert, J.
Antigenic cross-reactivity of major outer membrane proteins in enterobacteriaceae species.
J.
Gen.
Microbiol.
111 , 293–302 (1979).
Baker, C.
J.
& Kasper, D.
L.
Correlation of maternal antibody deficiency with susceptibility to neonatal group B streptococcal infection.
N.
Engl.
J.
Med.
294 , 753–756 (1976).
Chace, D.
H., De Jesus, V.
R.
& Spitzer, A.
R.
Clinical chemistry and dried blood spots: increasing laboratory utilization by improved understanding of quantitative challenges.
Bioanalysis 6 , 2791–2794 (2014).
Nhu, N.
T.
K.
High-risk Escherichia coli clones that cause neonatal meningitis and association with recrudescent infection.
eLife 12 , RP91853 (2024).
Cole, B.
K., Ilikj, M., McCloskey, C.
B.
& Chavez-Bueno, S.
Antibiotic resistance and molecular characterization of bacteremia Escherichia coli isolates from newborns in the United States.
PLoS ONE 14 , e0219352 (2019).
Malaure, C.
et al.
Early-onset infection caused by Escherichia coli sequence type 1193 in late preterm and full-term neonates.
Emerg.
Infect.
Dis.
30 , 20–28 (2024).
Mulvey, M.
A., Schilling, J.
D.
& Hultgren, S.
J.
Establishment of a persistent Escherichia coli reservoir during the acute phase of a bladder infection.
Infect.
Immun.
69 , 4572–4579 (2001).
Day, M.
W., Jackson, L.
A., Akins, D.
R., Dyer, D.
W.
& Chavez-Bueno, S.
Whole-genome sequences of the archetypal K1 Escherichia coli neonatal isolate RS218 and contemporary neonatal bacteremia clinical isolates SCB11, SCB12, and SCB15.
Genome Announc.
3 , e01598-14 (2015).
Williams, M.
et al.
Whole-genome sequencing-based phylogeny, antibiotic resistance, and invasive phenotype of Escherichia coli strains colonizing the cervix of women in preterm labor.
BMC Microbiol.
21 , 330 (2021).
Chavez-Bueno, S., Day, M.
W., Toby, I.
T., Akins, D.
R.
& Dyer, D.
W.
Genome sequence of SCB34, a sequence type 131 multidrug-resistant Escherichia coli isolate causing neonatal early-onset sepsis.
Genome Announc.
https://doi.org/10.1128/genomeA.00514-14 (2014).
Gu, H.
et al.
Rational design and evaluation of an artificial Escherichia coli K1 protein vaccine candidate based on the structure of OmpA.
Front.
Cell Infect.
Microbiol.
8 , 172 (2018).
Mayorga, M., Wheatley, J.
L., Maldonado, B.
N.
& Chavez-Bueno, S.
Bovine lactoferrin maintains antibacterial effect against neonatal Escherichia coli septicaemia isolates despite the presence of iron acquisition genes.
J.
Med.
Microbiol.
https://doi.org/10.1099/jmm.0.002116 (2026).
Sarkar, S., Ulett, G.
C., Totsika, M., Phan, M.
D.
& Schembri, M.
A.
Role of capsule and O antigen in the virulence of uropathogenic Escherichia coli .
PLoS ONE 9 , e94786 (2014).
Article ADS PubMed PubMed Central Google Scholar
Phan, M.
D.
et al.
The serum resistome of a globally disseminated multidrug resistant uropathogenic Escherichia coli clone.
PLoS Genet.
9 , e1003834 (2013).
Pautsch, A.
& Schulz, G.
E.
Structure of the outer membrane protein A transmembrane domain.
Nat.
Struct.
Biol.
5 , 1013–1017 (1998).
Netea, M.
G.
et al.
Defining trained immunity and its role in health and disease.
Nat.
Rev.
Immunol.
20 , 375–388 (2020).
Rusconi, B.
et al.
Intergenerational protective anti-gut commensal immunoglobulin G originates in early life.
Proc.
Natl Acad.
Sci.
USA 121 , e2309994121 (2024).
Cohen, I.
R.
& Norins, L.
C.
Antibiodies of the IgG, IgM, and IgA classes in newborn and adult sera reactive with gram-negative bacteria.
J.
Clin.
Invest.
47 , 1053–1062 (1968).
Crook, N.
et al.
Adaptive strategies of the candidate probiotic E.
coli Nissle in the mammalian gut.
Cell Host Microbe 25 , 499–512.e8 (2019).
Caballero-Flores, G.
et al.
Maternal immunization confers protection to the offspring against an attaching and effacing pathogen through delivery of IgG in breast milk.
Cell Host Microbe 25 , 313–323.e4 (2019).
Sanidad, K.
Z.
et al.
Maternal gut microbiome-induced IgG regulates neonatal gut microbiome and immunity.
Sci.
Immunol.
7 , eabh3816 (2022).
Smith, P., DiLillo, D.
J., Bournazos, S., Li, F.
& Ravetch, J.
V.
Mouse model recapitulating human Fcγ receptor structural and functional diversity.
Proc.
Natl Acad.
Sci.
USA 109 , 6181–6186 (2012).
Erickson, J.
J.
et al.
Pregnancy enables antibody protection against intracellular infection.
Nature 606 , 769–775 (2022).
Lawrence, S.
M., Corriden, R.
& Nizet, V.
Age-appropriate functions and dysfunctions of the neonatal neutrophil.
Front.
Pediatr.
5 , 23 (2017).
McGreal, E.
P., Hearne, K.
& Spiller, O.
B.
Off to a slow start: under-development of the complement system in term newborns is more substantial following premature birth.
Immunobiology 217 , 176–186 (2012).
Balogh, P., Tew, J.
G.
& Szakal, A.
K.
Simultaneous blockade of Fcγ receptors and indirect labeling of mouse lymphocytes by the selective detection of allotype-restricted epitopes on the kappa chain of rat monoclonal antibodies.
Cytometry 47 , 107–110 (2002).
Berry, G.
T.
Galactosemia: when is it a newborn screening emergency?
Mol.
Genet.
Metab.
106 , 7–11 (2012).
Xu, Y.
et al.
Antibiotic exposure prevents acquisition of beneficial metabolic functions in the preterm infant gut microbiome.
Microbiome 10 , 103 (2022).
Hwang, J.
et al.
Predictive factors for perinatal bacterial transmission from colonized mothers to delivered very-low-birth-weight infants: a retrospective cohort study.
Sci.
Rep.
14 , 16835 (2024).
Malek, A., Sager, R., Kuhn, P., Nicolaides, K.
H.
& Schneider, H.
Evolution of maternofetal transport of immunoglobulins during human pregnancy.
Am.
J.
Reprod.
Immunol.
36 , 248–255 (1996).
Zhang, S.
L.
et al.
Maternal and neonatal IgG against Klebsiella pneumoniae are associated with lower risk of neonatal sepsis: a case-control study of hospitalized neonates in Botswana.
PLOS Glob.
Public Health 4 , e0003350 (2024).
Madhi, S.
A.
et al.
Influenza vaccination of pregnant women and protection of their infants.
N.
Engl.
J.
Med.
371 , 918–931 (2014).
Pasetti, M.
F.
et al.
Maternal determinants of infant immunity: Implications for effective immunization and maternal-child health.
Vaccine 38 , 4491–4494 (2020).
Atyeo, C.
G.
et al.
Maternal immune response and placental antibody transfer after COVID-19 vaccination across trimester and platforms.
Nat.
Commun.
13 , 3571 (2022).
Kollmann, T.
R., Marchant, A.
& Way, S.
S.
Vaccination strategies to enhance immunity in neonates.
Science 368 , 612–615 (2020).
Qiu, L.
et al.
Vaccines against extraintestinal pathogenic Escherichia coli (ExPEC): progress and challenges.
Gut Microbes 16 , 2359691 (2024).
Wessels, M.
R.
et al.
Studies of group B streptococcal infection in mice deficient in complement component C3 or C4 demonstrate an essential role for complement in both innate and acquired immunity.
Proc.
Natl Acad.
Sci.
USA 92 , 11490–11494 (1995).
Fonseca, M.
I.
et al.
Cell-specific deletion of C1qa identifies microglia as the dominant source of C1q in mouse brain.
J.
Neuroinflammation 14 , 48 (2017).
Kitamura, D., Roes, J., Kuhn, R.
& Rajewsky, K.
A B cell-deficient mouse by targeted disruption of the membrane exon of the immunoglobulin mu chain gene.
Nature 350 , 423–426 (1991).
Nitschke, L., Carsetti, R., Ocker, B., Kohler, G.
& Lamers, M.
C.
CD22 is a negative regulator of B-cell receptor signalling.
Curr.
Biol.
7 , 133–143 (1997).
Totsika, M.
et al.
Insights into a multidrug resistant Escherichia coli pathogen of the globally disseminated ST131 lineage: genome analysis and virulence mechanisms.
PLoS ONE 6 , e26578 (2011).
Nhu, N.
T.
K.
et al.
Discovery of new genes involved in Curli production by a uropathogenic escherichia coli strain from the highly virulent O45:K1:H7 lineage.
mBio https://doi.org/10.1128/mBio.01462-18 (2018).
Mobley, H.
L.
et al.
Pyelonephritogenic Escherichia coli and killing of cultured human renal proximal tubular epithelial cells: role of hemolysin in some strains.
Infect.
Immun.
58 , 1281–1289 (1990).
Nhu, N.
T.
K.
et al.
A convergent evolutionary pathway attenuating cellulose production drives enhanced virulence of some bacteria.
Nat.
Commun.
15 , 1441 (2024).
Choi, J., Schmukler, M.
& Groisman, E.
A.
Degradation of gene silencer is essential for expression of foreign genes and bacterial colonization of the mammalian gut.
Proc.
Natl Acad.
Sci.
USA 119 , e2210239119 (2022).
Budnick, J.
A., Bina, X.
R.
& Bina, J.
E.
Complete genome sequence of Klebsiella pneumoniae strain ATCC 43816.
Microbiol.
Resour.
Announc.
https://doi.org/10.1128/MRA.01441-20 (2021).
Gort, A.
S.
& Miller, V.
L.
Identification and characterization of Yersinia enterocolitica genes induced during systemic infection.
Infect Immun 68 , 6633–6642 (2000).
Montano, J.
et al.
Salmonella enterica Serovar Typhimurium 14028s genomic regions required for colonization of lettuce leaves.
Front.
Microbiol.
11 , 6 (2020).
Becavin, C.
et al.
Comparison of widely used Listeria monocytogenes strains EGD, 10403S, and EGD-e highlights genomic variations underlying differences in pathogenicity.
mBio 5 , e00969–00914 (2014).
Ren, Y.
et al.
Complete genome sequence of Enterobacter cloacae subsp.
cloacae type strain ATCC 13047.
J.
Bacteriol.
192 , 2463–2464 (2010).
Shao, T.
Y.
et al.
Commensal Candida albicans positively calibrates systemic Th17 immunological responses.
Cell Host Microbe 25 , 404–417.e6 (2019).
Prasanphanich, N.
S.
et al.
Preconceptual priming overrides susceptibility to Escherichia coli systemic infection during pregnancy.
mBio https://doi.org/10.1128/mBio.00002-21 (2021).
Jiang, T.
T.
et al.
Commensal fungi recapitulate the protective benefits of intestinal bacteria.
Cell Host Microbe 22 , 809–816.e4 (2017).
Wimmers, F.
et al.
Multi-omics analysis of mucosal and systemic immunity to SARS-CoV-2 after birth.
Cell 186 , 4632–4651.e23 (2023).
Langbo, C., Bach, J., Kleyn, M.
& Downes, F.
P.
From newborn screening to population health research: implementation of the Michigan BioTrust for health.
Public Health Rep.
128 , 377–384 (2013).
Mei, J.
V., Alexander, J.
R., Adam, B.
W.
& Hannon, W.
H.
Use of filter paper for the collection and analysis of human whole blood specimens.
J.
Nutr.
131 , 1631S–1636S (2001).
Mercader, S., Featherstone, D.
& Bellini, W.
J.
Comparison of available methods to elute serum from dried blood spot samples for measles serology.
J.
Virol.
Methods 137 , 140–149 (2006).
Gruner, N., Stambouli, O.
& Ross, R.
S.
Dried blood spots–preparing and processing for use in immunoassays and in molecular techniques.
J.
Vis.
Exp.
97 , e52619 (2015).
Lee, T.
S.
et al.
BglBrick vectors and datasheets: a synthetic biology platform for gene expression.
J.
Biol.
Eng.
5 , 12 (2011).
Hegde, S.
et al.
Inhibition of RHOA activity preserves the survival and hemostasis function of long-term cold-stored platelets.
Blood 144 , 1732–1746 (2024).
Millius, A.
& Weiner, O.
D.
Manipulation of neutrophil-like HL-60 cells for the study of directed cell migration.
Methods Mol.
Biol.
591 , 147–158 (2010).
De Coster, W., D’Hert, S., Schultz, D.
T., Cruts, M.
& Van Broeckhoven, C.
NanoPack: visualizing and processing long-read sequencing data.
Bioinformatics 34 , 2666–2669 (2018).
Bankevich, A.
et al.
SPAdes: a new genome assembly algorithm and its applications to single-cell sequencing.
J.
Comput.
Biol.
19 , 455–477 (2012).
Article MathSciNet CAS PubMed PubMed Central Google Scholar
Vaser, R., Sovic, I., Nagarajan, N.
& Sikic, M.
Fast and accurate de novo genome assembly from long uncorrected reads.
Genome Res.
27 , 737–746 (2017).
Wick, R.
R., Judd, L.
M., Gorrie, C.
L.
& Holt, K.
E.
Unicycler: resolving bacterial genome assemblies from short and long sequencing reads.
PLoS Comput.
Biol.
13 , e1005595 (2017).
Treangen, T.
J., Ondov, B.
D., Koren, S.
& Phillippy, A.
M.
The Harvest suite for rapid core-genome alignment and visualization of thousands of intraspecific microbial genomes.
Genome Biol.
15 , 524 (2014).
Deatherage, D.
E.
& Barrick, J.
E.
Identification of mutations in laboratory-evolved microbes from next-generation sequencing data using breseq.
Methods Mol.
Biol.
1151 , 165–188 (2014).
Katoh, K.
& Standley, D.
M.
MAFFT multiple sequence alignment software version 7: improvements in performance and usability.
Mol.
Biol.
Evol.
30 , 772–780 (2013).
We thank Cincinnati Children’s Hospital Comparative Medicine division led by S.
Tummala for animal care; J.
V.
Ravetch for providing FcγR null (FcRα null) mice; the Michigan Department of Health and Human Services Medicaid, Newborn Screening, and BioTrust programme staff for selecting and preparing dried blood spot samples and associated data for newborn screening cases and control babies; L.
Ray, J.
Crawford and C.
Lutzko of Cincinnati Children’s Hospital Cellular Manipulations Laboratory and Cell Processing Facility for providing de-identified human cord blood specimens; and S.
G.
Kallapur and Y.
Zheng and the Center for Inflammation and Tolerance faculty for helpful discussions.
This work was supported in part by NIH-NIAID through grants R01AI175431, R01AI189741, R01AI184537 (S.S.W.), U01AI144673 (M.
A.
Staat), Good Ventures Foundation (M.
A.
Staat), the Burroughs Wellcome Fund Investigator in the Pathogenesis of Infectious Disease Award (grant 1011031, to S.S.W.), March of Dimes Ohio Prematurity Research Collaborative (S.S.W.), National Health and Medical Research Council (NHMRC) grants 1181958, 2001431 (M.
A.
Schembri, M.-D.P.
and N.T.K.N.) and 2037698 (M.
A.
Schembri and M.-D.P.).
Division of Infectious Diseases, Center for Inflammation and Tolerance, Cincinnati Children’s Hospital Department of Pediatrics, University of Cincinnati College of Medicine, Cincinnati, OH, USA
Raymond E.
Diep, Ujjwal Adhikari, Giang Pham, Allison R.
Burrell, Mary A.
Staat, David B.
Haslam & Sing Sing Way
Division of Neonatology, Center for Inflammation and Tolerance, Cincinnati Children’s Hospital Department of Pediatrics, University of Cincinnati College of Medicine, Cincinnati, OH, USA
Kubra Gokce Tezel & John J.
Erickson
Institute for Molecular Bioscience (IMB), The University of Queensland, Brisbane, Queensland, Australia
Nguyen Thi Khanh Nhu, Minh-Duy Phan, Kate M.
Peters & Mark A.
Schembri
Australian Infectious Diseases Research Centre, The University of Queensland, Brisbane, Queensland, Australia
School of Chemistry and Molecular Biosciences, The University of Queensland, Brisbane, Queensland, Australia
Lyda Hill Department of Bioinformatics, University of Texas Southwestern Medical Center, Dallas, TX, USA
Division of Infectious Diseases, Department of Pediatrics, Children’s Mercy Kansas City and University of Missouri Kansas City School of Medicine, Kansas City, MO, USA
Search author on: PubMed Google Scholar
M.
A.
Staat, D.B.H., J.J.E., S.C.-B., M.
A.
Schembri and S.S.W.
designed research.
R.E.D., U.A., K.G.T., G.P., N.T.K.N., M.-D.P.
and K.M.P.
conducted experiments.
R.E.D., K.G.T., S.H.S., D.B.H., J.J.E., S.C.-B.
and S.S.W.
analysed data.
A.R.B., M.
A.
Staat and M.
A.
Schembri provided essential reagents.
S.S.W.
acquired funding.
All authors wrote and edited the manuscript.
Correspondence to Sing Sing Way .
The authors declare no competing interests.
Nature thanks Gabriel Nunez, Phillip Tarr and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Peer review reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig.
1 E.
coli Nissle (EcN) colonization-induced systemic immunogenicity cross-reactive to neonatal infection clinical isolates.
a , EcN levels in each tissue 50 days after a single oral EcN inoculation administered to 6–8 week old WT adult mice on the C57BL/6 background reared under specific pathogen free laboratory conditions (n = 6).
b , anti-EcN IgG end point (EcEPT) titres in the sera of mice 50 days after EcN oral inoculation (n = 8) compared with control mice without E.
coli colonization (n = 7) using three independently prepared batches of EcN coated ELISA plates.
c , anti-EcN titres for each isotype in the sera of mice 50 days after EcN oral inoculation (n = 7–10 mice, with each dot representing the data from an individual animal) compared with control mice without E.
coli colonization (n = 7–10 mice, with each dot representing the data from an individual animal).
d , Sequence type, O-antigen, K-capsule and flagella serotype for each E.
coli clinical isolate.
e , IgG end point (EcEPT) titres against each E.
coli strain described in panel d in the sera of adult mice 50 days after a single oral EcN inoculation (n = 8–14 mice/group, with each dot representing the data from an individual animal) compared with no E.
coli colonization control mice (n = 5–11 mice/group, with each dot representing the data from an individual animal).
Data are presented as mean values (bar) ± one standard deviation.
Differences between groups analysed using unpaired Student’s t test.
LOD, limits of detection.
Extended Data Fig.
2 E.
coli Nissle (EcN) colonization primes serological cross-reactivity to other strains with individual or combined deficiency in O-antigen, K-capsule, and flagella production.
IgG end point (EcEPT) titres against each E.
coli strain in the sera of adult mice 50 days after a single oral EcN inoculation (n = 8) compared with no E.
coli colonization control mice (n = 7).
Data are presented as mean values (bar) ± one standard deviation.
Differences between groups analysed using unpaired Student’s t test.
Extended Data Fig.
3 Commensal E.
coli Nissle (EcN) primed immunogenicity to OmpA outer loop peptides.
a , Schematic highlighting eight transmembrane domains at the N-terminus of E.
coli OmpA resulting in four distinct outer facing loop peptides (not to scale).
b , Sequence alignment for each OmpA outer loop peptide across multiple E.
coli strains, highlighting two separate loop 3 variants, and IgG titres against each peptide in the sera of adult mice 50 days after oral inoculation with EcN (n = 10) or Δ ompA EcN (n = 10) compared with control mice without E.
coli colonization (n = 10).
c , EcN or Δ ompA EcN CFUs in the feces after a single oral inoculation (2 × 10 6 CFUs each strain) to 6–8 week old C57BL/6 WT mice under specific pathogen free laboratory conditions.
Data are presented as mean values (bar) ± one standard deviation.
Differences between groups analysed using one-way ANOVA.
Diagrams in panel a created using BioRender; Way, S.
S.
https://biorender.com/wz9mo0w (2026).
Extended Data Fig.
4 IgG cross-reactivity against OmpA-deficient E.
coli Nissle (EcN) compared to other clinical isolates.
a , Table showing sequence type, O-antigen, K-capsule and flagella serotype for each E.
coli clinical isolate.
b , IgG end point EcEPT titres against each WT or OmpA-deficient isogenic strain in the serum of mice 50 days after oral EcN inoculation (n = 8–12 mice/group, with each dot representing the data from an individual animal) compared with control mice without E.
coli colonization (n = 7–11 mice/group) (left), and fold-increase in IgG titres in the serum of EcN colonized compared with no E.
coli colonization control mice against each strain (right).
Data are presented as mean values (bar) ± one standard deviation.
Differences between groups analysed using unpaired Student’s t test.
Extended Data Fig.
5 Commensal E.
coli Nissle (EcN) primed immunogenicity cross-reactive to other Enterobacteriaceae.
a , IgG end point EcEPT titres against each bacterial or fungal species in the serum of adult mice 50 days after oral EcN inoculation (n = 7–8 mice/group, with each dot representing the data from an individual animal) compared with control mice without E.
coli colonization (n = 6–7 mice/group, with each dot representing the data from an individual animal).
b , Sequence alignment for each OmpA outer loop peptide across multiple E.
coli strains or Enterobacteriaceae species.
Data are presented as mean values (bar) ± one standard deviation.
Differences between groups analysed using unpaired Student’s t test.
Extended Data Fig.
6 EcN colonization induced protection against systemic infection requires B cells.
a , Bacterial burden one day after SCB61 or UTI89 infection for mice 50 days after EcN colonization (n = 8) compared with no colonization control mice (n = 7 [SCB61]; n = 8 [UTI89]).
b , Bacterial burden one day after UTI89 infection for mice 50 days after oral inoculation with EcN (n = 8) or Δ ompA EcN (n = 8) compared with control mice without E.
coli colonization (n = 8).
c , Anti- E.
coli UTI89 IgG titers for mice 50 days after oral inoculation with EcN (n = 10) or Δ ompA EcN (n = 10) compared with control mice without E.
coli colonization (n = 10).
d , Klebsiella pneumoniae CFUs one day after infection (10 5 CFUs, strain 43816 administered IV) for mice 50 days after EcN colonization (n = 8) compared with no colonization control mice (n = 8).
e , Listeria monocytogenes CFUs one day after infection (10 5 CFUs, strain 10403 s administered IV) for mice 50 days after EcN colonization (n = 8) compared with no colonization control mice (n = 7).
f , Staphylococcus aureus CFUs one day after infection (7.5 × 10 6 CFUs, strain USA200 administered IV) for mice 50 days after EcN colonization (n = 7) compared with no colonization control mice (n = 8).
g , Candida albicans CFUs one day after infection (10 6 CFUs, strain SC5314 administered IV) for mice 50 days after EcN colonization (n = 8) compared with no colonization control mice (n = 7).
h , Bacterial burden one day after SCB61 or UTI89 infection for mice administered sera (100 μl) from control mice without EcN colonization (n = 8), WT mice day 50 after EcN colonization (n = 7 [SCB61]; n = 8 [UTI89]) or μMT -/- mice day 50 after EcN colonization (n = 7 [SCB61]; n = 8 [UTI89]).
i , Bacterial burden one day after UTI89 infection in WT compared with μMT -/- mice day 50 after EcN colonization (n = 12 [WT]; n = 13 [ μMT -/-]) compared with no colonization controls (n = 12 [WT]; n = 11 [ μMT -/-]).
j , EcN levels in the feces at each time point (left) or day 50 (right) after a single oral inoculation to WT (n = 12) or μMT -/- (n = 10) mice.
Data are presented as mean values (bar) ± one standard deviation.
Differences between groups analysed using unpaired Student’s t test (panels a, d - g, i, j) or one-way ANOVA (panels b, c, h).
LOD, limits of detection.
Extended Data Fig.
7 Natural anti- E.
coli maternal antibodies override neonatal infection susceptibility.
a , Survival and bacterial burden one day after infection for 8-week (adult) (n = 6) compared with 3-day (neonate) (n = 7) old mice intraperitoneally (IP) infected with 200 CFUs E.
coli UTI89.
b , Survival and bacterial burden one day after UTI89 infection (200 CFUs, administered IP) in 3-day old pups born to control mice without EcN colonization (n = 9) compared with EcN colonized WT (n = 10) or EcN colonized μMT -/- (n = 7) dams.
c , Survival and bacterial burden one day after UTI89 infection (5 × 10 6 CFUs, oral gavage) in 3-day old pups born to control mice without EcN colonization (n = 10) compared with EcN colonized WT (n = 9) dams.
d , Anti-EcN IgG or IgM titers in EcN colonized dams and their 3-day old pups (n = 5; lines indicate each paired maternal:pooled pup specimens).
e , Bacterial burden one day after UTI89 infection (200 CFUs, administered IP) in 3-day old pups administered each dosage of purified IgG from EcN colonized or control mice without E.
coli colonization one hour prior to infection (n = 3–5 mice/group, with each dot representing the data from an individual animal).
f , Nucleotide differences between two parental EcN stocks, compared with strains isolated from the feces of EcN colonized virgin or postpartum mice.
g , Bacterial burden one day after UTI89 infection (200 CFUs, administered IP) (left) or compared with anti-EcN IgG titers (right) for pups born to EcN colonized dams (n = 10), pups born to control mice without EcN colonization (n = 9) or cross-fostered within the first 12 h after birth (n = 8 [prenatal]; n = 9 [postnatal]).
Data are presented as mean values (bar) ± one standard deviation.
Differences between groups analysed using unpaired Student’s t test (panels a, c), one-way ANOVA (panels b, g [left]), paired Student’s t test (panel d), using Log-rank (Mantel-Cox) test (panels a - c), or simple linear regression (panel g [right]).
LOD, limits of detection.
Extended Data Fig.
8 Paired human maternal-cord blood specimens demonstrating anti- E.
coli IgG vertical transfer, and the necessity for FcγR and complement for natural antibody conferred protection against E.
coli infection.
a , Demographical and pregnancy outcome data for each maternal-infant pair.
b , Anti-EcN titers for IgM and each IgG subclass in paired maternal-cord blood serum specimens (n = 10; lines indicate each paired maternal:cord blood specimens).
c , IgG titers against each OmpA outer loop peptide for each paired maternal-cord blood serum specimens (n = 10; lines indicate each paired maternal:cord blood specimens).
d , E.
coli CFUs in the spleen after UTI89 infection (200 CFUs) in 3-day old pups administered sera from control mice without EcN colonization (n = 9) compared with sera from EcN colonized WT (n = 5) or EcN colonized μMT -/- (n = 8) mouse donors.
e , E.
coli CFUs in the spleen after UTI89 infection in 3-day old pup or 8-week old adult FcγR null , C3 -/-, C1q -/- or Cd22 -/- recipients administered sera from EcN colonized WT donor mice (n = 6–8 mice/group, with each dot representing the data from an individual animal) compared with sera from control mice without EcN colonization (n = 6–8 mice/group, with each dot representing the data from an individual animal).
Data are presented as mean values (bar) ± one standard deviation.
Differences between groups analysed using paired Student’s t test (panels b, c), one-way ANOVA (panel d) or unpaired Student’s t test (panel e).
LOD, limits of detection.
Extended Data Fig.
9 Comparable gestational age dependent changes in total IgG levels in newborn blood spot specimens regardless of neonatal sepsis.
a , Anti- E.
coli end point (EcEPT) titers for each IgG subclass in newborn blood specimens from babies with E.
coli sepsis (n = 100) compared with the average of controls (n = 100) matched for infant sex, gestational age, and birth timing titered using a mixture of eight neonatal infection clinical isolates (RS218, SCB12, SCB29, SCB61, SCB34, SCB58, SCB37, SCB60).
b , c , Levels of total IgG or each IgG subclass from newborn blood spot specimens of babies with E.
coli sepsis (n = 100) compared with the average of controls (n = 100; panel b) or each control (n = 296; panel c) matched for infant sex, gestational age, and birth timing (panel b) or based on sex and gestational age (panel c).
Triangle, female infants; square, male infants.
Differences between groups analysed using paired Student’s t test (panels a, b) or simple linear regression (panel c).
LOD, limits of detection.
Extended Data Fig.
10 Diminished anti- E.
coli IgG and opsonization activity in newborn blood spots of babies with E.
coli sepsis.
a–d , Anti- E.
coli IgG levels in blood spots from babies with E.
coli sepsis (n = 100) and matched controls (n = 296) titered against pooled neonatal clinical isolates (RS218, SCB12, SCB29, SCB61, SCB34, SCB58, SCB37, SCB60) (a), titered against pooled OmpA outer loop peptides (b), opsonization activity against eight pooled E.
coli clinical isolates in THP1 macrophage (c) or HL60 neutrophil cells (d) (left), and probability of E.
coli sepsis for each parameter using conditional logistic regression accounting for 1:3 case-control matching (anti- E.
coli IgG, anti-OmpA IgG, and anti- E.
coli opsonization THP1 cells), or standard logistic regression for anti- E.
coli opsonization HL60 cells where separation between matched cases and controls was sufficient to prevent convergence of conditional logistic regression, with shading indicating 95% confidence intervals (right).
Data are presented as mean values (bar) ± one standard deviation.
e , Anti- E.
coli IgG titres in blood spots from babies with E.
coli sepsis (n = 100) compared with the average in matched control babies with infection (n = 100) titered against pooled eight neonatal clinical isolates evaluated by infection onset timing.
f , IgG levels in blood spots of babies described in panel e (n = 100 [ E.
coli sepsis]; n = 100 [averaged control babies without infection]).
g , h , IgG titers against in blood spots against pooled OmpA loop peptides (g) or scramble control peptides (h) for babies described in panel e (n = 100 [ E.
coli sepsis]; n = 100 [averaged control babies without infection]).
i , j , Anti- E.
coli opsonization activity in blood spot specimens against eight pooled E.
coli clinical isolates in THP1 macrophage (i) or HL60 neutrophil cells (j) for babies described in panel e (n = 100 [ E.
coli sepsis]; n = 100 [averaged control babies without infection]).
Each point represents the data from individual blood spot specimens of E.
coli sepsis cases (red) or the average from 3 matched controls for each case (blue) with 95% confidence intervals highlighted.
k , Anti- E.
coli or anti-OmpA IgG titres, or anti- E.
coli opsonization activity in THP1 macrophage or HL60 neutrophil cells of blood spots of babies identified with E.
coli sepsis within the first 2 days after birth (n = 44) or later onset infection (n = 56).
Differences between groups analysed using unparied (panel a-d) or paired Student’s t test (panel k), and between regression intercepts using Analysis of Covariance (e - j).
Reporting Summary (download PDF )
Peer Review File (download PDF )
Source Data Fig.
1 (download XLSX )
Source Data Fig.
2 (download XLSX )
Source Data Fig.
3 (download XLSX )
Source Data Fig.
4 (download XLSX )
Source Data Extended Data Fig.
1 (download XLSX )
Source Data Extended Data Fig.
2 (download XLSX )
Source Data Extended Data Fig.
3 (download XLSX )
Source Data Extended Data Fig.
4 (download XLSX )
Source Data Extended Data Fig.
5 (download XLSX )
Source Data Extended Data Fig.
6 (download XLSX )
Source Data Extended Data Fig.
7 (download XLSX )
Source Data Extended Data Fig.
8 (download XLSX )
Source Data Extended Data Fig.
9 (download XLSX )
Source Data Extended Data Fig.
10 (download XLSX )
Version of record : 11 March 2026
DOI : https://doi.org/10.1038/s41586-026-10225-z
Related Stories
Source: This article was originally published by Nature News
Read Full Original Article →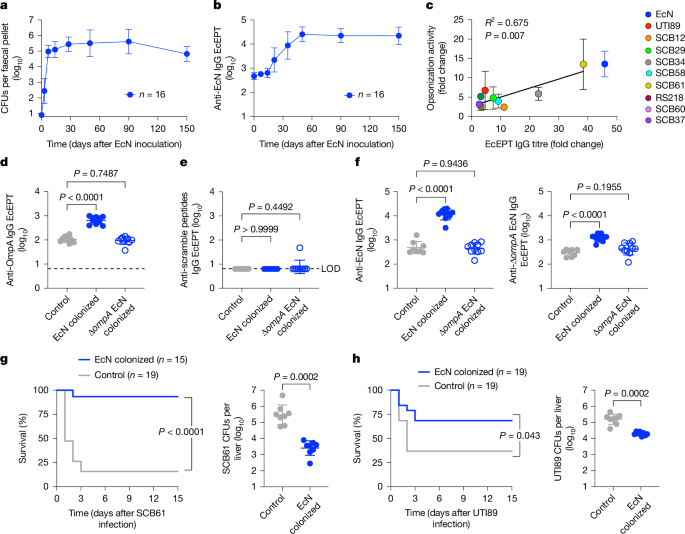



Comments (0)
No comments yet. Be the first to comment!
Leave a Comment